Around 8 to 12 weeks after stopping a GLP-1 medication, the biology that made food feel manageable begins to shift. The hunger signals that were quieted start to re-emerge. Cravings that had been essentially offline return – sometimes with an urgency that surprises people who spent months barely thinking about food.
This is not a willpower failure. This is biology doing exactly what it was designed to do.
But here is what the research is starting to make clear: not everyone experiences this the same way. Some people discontinue GLP-1 therapy and maintain the majority of their weight loss. Others regain most – or all – of it within 12 months. The difference between these two groups is increasingly predictable, and it maps onto four measurable variables that together form what clinicians are beginning to call a relapse prediction model.
Understanding where you fall in this model is arguably the most important thing you can do while still on the medication.
What ‘Relapse’ Means in GLP-1 Biology
Before covering the model itself, it helps to reframe what relapse actually means in this context.
When clinicians talk about weight relapse after GLP-1 discontinuation, they are not describing a moral or motivational failure. They are describing a physiological recalibration – the body’s attempt to return to a defended weight setpoint that was suppressed, but not reset, by the medication.
GLP-1 receptor agonists work primarily by mimicking a gut hormone that signals satiety to the brain, slowing gastric emptying, and modulating reward circuits associated with food. What they do not do – at least not directly – is reprogram the underlying neural architecture that determines baseline drive toward food. That architecture, shaped by genetics, stress history, sleep patterns, and years of eating behavior, remains largely in place beneath the medication’s effect.
When the drug leaves the system, that architecture re-emerges. The question is not whether it will – it will – but how much influence it has over behavior, and how much structural support you have built to counterbalance it.
The STEP 1 trial extension confirmed this pattern clearly. One year after withdrawal of once-weekly semaglutide 2.4 mg, participants regained two-thirds of their prior weight loss, with cardiometabolic improvements also reverting toward baseline.[1] A 2026 editorial reviewing the STEP 4 and SURMOUNT-4 withdrawal data described post-discontinuation weight regain as “disease recurrence” rather than therapeutic failure – a reframing that carries real clinical implications for how these medications are understood and used.[2]
The 4-Factor Relapse Prediction Model
Factor 1: Neural Reward Sensitivity
The brain’s dopaminergic reward system is not static. In people with high baseline reward sensitivity – characterized by stronger craving responses, greater reactivity to food cues, and a history of emotional or stress-driven eating – GLP-1 medications tend to produce more dramatic short-term results, but also greater vulnerability after discontinuation.
This is because the medication was doing a larger share of the regulatory work. When it is removed, the gap between baseline drive and behavioral capacity is wider.
Research using fMRI food-cue paradigms has shown that the interaction between circulating endogenous GLP-1 and activity in the dorsolateral prefrontal cortex – a region involved in behavioral control – is a strong predictor of long-term body weight change. Neither GLP-1 signaling nor prefrontal activity alone predicted outcomes; their concurrent activation was what mattered.[3] This points to why the medication period is also a window for building cognitive regulatory habits, not just metabolic ones.
How to self-assess: Honest reflection on pre-medication patterns is useful here. Did food function as a primary stress regulator? Were cravings largely cue-driven rather than hunger-driven? High neural reward sensitivity means behavioral scaffolding work (Factor 3 below) needs to be proportionally more robust.
Factor 2: Gut-Brain Axis Recalibration Status
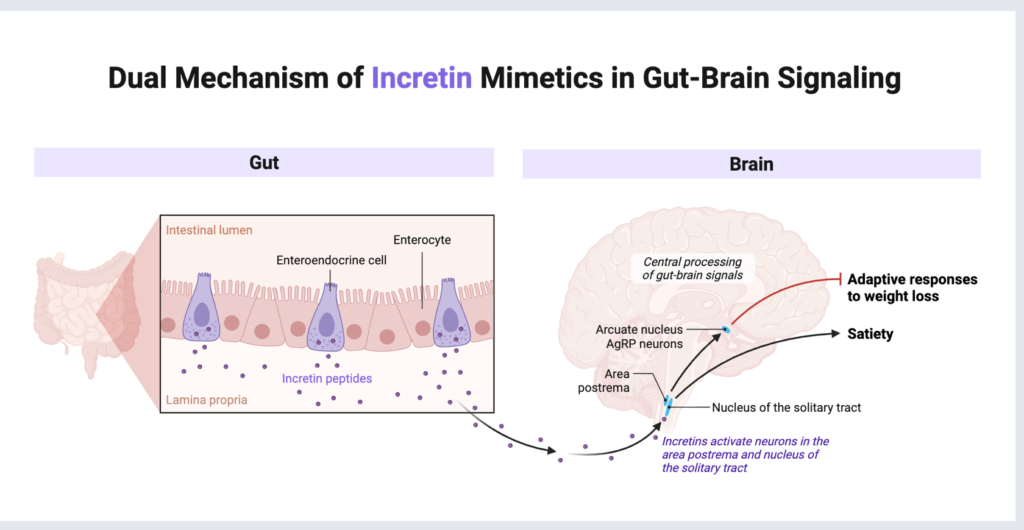
The gut-brain axis – the bidirectional communication network between the gastrointestinal system and the central nervous system – is profoundly influenced by dietary patterns and gut microbial composition. GLP-1 medications interact directly with this axis, but they do not permanently alter it.
Research suggests that people who use the medication period to meaningfully shift their dietary patterns – toward higher fiber, more diverse plant intake, lower ultra-processed food load – show better recalibration of endogenous GLP-1 production and gut signaling over time. In practical terms, the biology becomes more capable of generating satiety signals independently.
How to self-assess: Have dietary patterns genuinely shifted during the medication period, or have previous habits largely continued with reduced portions? The former suggests improving gut-brain recalibration. The latter is a meaningful risk signal.
Factor 3: Behavioral Scaffolding Depth
This is the most modifiable factor, and the one that most directly predicts outcomes at the 12-month mark after discontinuation.
Behavioral scaffolding refers to the collection of structured habits, environmental modifications, and relational supports that make healthy behavior automatic rather than effortful. Think of it as infrastructure. When pharmacological support is removed, the infrastructure either holds the new pattern in place or it does not.
High scaffolding depth looks like: consistent meal timing, reliable sleep architecture, a defined movement practice, social environments that support rather than undermine eating behavior, and a clear plan for what happens when cravings intensify.
Low scaffolding depth looks like: good intentions and some general awareness, but no specific structures. Relying on motivation and willpower as the primary regulatory mechanism.
A study evaluating appetite hormones and eating behaviors as predictors of weight loss maintenance with GLP-1 therapy found that leptin response to meals – a proxy for satiety signaling capacity – was among the significant predictors of weight maintenance outcomes, while behavioral factors showed complex and context-dependent relationships with long-term results.[4] The takeaway is that no single biomarker determines outcomes; behavioral structure interacts with physiology throughout.
How to self-assess: If the medication were removed tomorrow, what specific structures would remain to support behavior? The specificity of your answer predicts your scaffolding score.
Factor 4: Metabolic Memory Signature
This is the most biologically complex factor and the one currently generating the most research interest.
Metabolic memory refers to epigenetic and cellular changes in adipose tissue, skeletal muscle, and the hypothalamus that persist after periods of obesity – and that create a biological pull back toward a previously held weight. People who have experienced multiple significant weight loss and regain cycles carry a stronger metabolic memory signature, meaning biological resistance to maintaining a lower weight is more entrenched.
A 2025 systematic review and meta-analysis found that weight recurrence following semaglutide discontinuation exceeded 50% of greatest on-treatment weight loss within 12 to 24 weeks across studies, with one cohort reaching 74.3% recurrence of lost weight by end of observation.[5] This is not a failure of character – it is a documented biological tendency that can be partially offset with specific strategies.
How to self-assess: A history of significant weight cycling – particularly cycles involving rapid loss followed by full regain – suggests an elevated metabolic memory signature. This factor is best addressed by prioritizing resistance training and protein adequacy during the medication period rather than maximum caloric restriction.
Reading Your Risk Profile
These four factors interact rather than operating in isolation. Someone with high neural reward sensitivity and low behavioral scaffolding is at significant relapse risk. Someone with improving gut-brain recalibration and high behavioral scaffolding may maintain effectively even with moderate reward sensitivity and metabolic memory concerns. The model is directional, not deterministic.
What it offers is a framework for allocating attention while still on the medication. The drug creates a window. The window is real and finite. What you build inside that window shapes the trajectory after it closes.
The Protective Window: What to Build Now
If you are currently on a GLP-1 medication – or considering starting one – this section is the most practically useful.
The medication period is not primarily about the weight lost during it. It is about what you build while the biological noise is quieted.
For neural reward sensitivity: Use the reduced food preoccupation to practice noticing hunger and satiety cues without acting on every food thought. This builds interoceptive literacy that matters when cravings return.
For gut-brain recalibration: Shift dietary patterns toward genuine food diversity and away from ultra-processed foods. The goal is to change what your gut microbiome is adapted to – not just what you eat less of.
For behavioral scaffolding: Build one structure at a time. Meal timing before movement routines. Sleep before meal composition. Scaffolding built incrementally is more durable than scaffolding built all at once.
For metabolic memory: Prioritize protein intake and resistance training. The evidence is clear that body composition during weight loss – not just the number on the scale – predicts long-term metabolic outcomes. Muscle is metabolically protective in ways that caloric restriction alone cannot replicate.
A Final Note on the Model
The relapse prediction model is not a reason for pessimism. It is a map.
Most of the variables in it are modifiable. The medication gives you leverage you have not had before on your own biology. The question worth sitting with is: what are you using that leverage to build?
The data consistently shows that the people who fare best after GLP-1 discontinuation are not the ones who lost the most weight during it. They are the ones who used the quieter biological environment to construct something that could stand without pharmacological support.
That is the work. And it is entirely possible.
Join the Field Notes
Metabolic Field Notes tracks the evidence as it develops, without protocol-pushing and without hype. If evidence-aware, translational science is useful to you, join the Field Notes.
REFERENCES
Based on articles retrieved from PubMed:
[1] Wilding JPH, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes, Obesity and Metabolism. 2022. https://doi.org/10.1111/dom.14725
[2] Quimbayo-Cifuentes AF. Weight Regain After GLP-1-Based Therapy Discontinuation: Failure, Physiology, or Follow-Up Gap. Cureus. 2026. https://doi.org/10.7759/cureus.104259
[3] Maurer L, et al. Interaction of circulating GLP-1 and the response of the dorsolateral prefrontal cortex to food-cues predicts body weight development. Molecular Metabolism. 2019. https://doi.org/10.1016/j.molmet.2019.08.014
[4] Bensignor MO, et al. Evaluating appetite/satiety hormones and eating behaviours as predictors of weight loss maintenance with GLP-1RA therapy in adolescents with severe obesity. Pediatric Obesity. 2024. https://doi.org/10.1111/ijpo.13105
[5] Garbaccio NC, et al. Plastic Surgery in the Ozempidemic: Considerations for the Timing of Body Contouring Surgery in Patients with Semaglutide-Associated Weight Loss. Aesthetic Plastic Surgery. 2025. https://doi.org/10.1007/s00266-025-05112-3
Medical Disclaimer: The content on this blog is for informational and educational purposes only and does not constitute professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.


